When in search of the truth about any topic it is always advisable to follow the trail of money. At least in our capitalist society. Unfortunately, this also holds true for the public health sector, where health should undoubtedly stand before financial interests.
The heavily promoted Gardasil® vaccine is supposed to prevent women from getting vulvar and vaginal cancer and provide a protection against diseases caused by the Human Papillomavirus (HPV) Types 6, 11, 16, and 18[1]. However, the vaccine has been subject to much criticism and is leaving a particularly interesting trail.
Complaints to the European Commission
Following numerous complaints of adverse effects experienced by women and girls shortly after receiving the HPV vaccine, Danish Health and Medicines Authorities (DHMA) had asked the European Medicines Agency (EMA) to review possible links of the HP-vaccine to functional disorders[2]. However, the official EMA report rejected any such links, whereby just months earlier an unofficial internal report (that later provided the basis for the official version) indeed showed that “a causal relationship between the dizziness and fatigue syndrome, Postural Orthostatic Tachycardia Syndrome (POTS) and Gardasil® [one of the HPV vaccines] can neither be confirmed nor denied”.[3]
The Nordic Cochrane Center has openly criticised the official whitewashing of the vaccines by drafting an open letter to the EMA that reads: “We find that the EMA’s comments are unprofessional, misleading, inappropriate and pejorative, and that the EMA’s approach involves cherry-picking, which is unscientific.” Furthermore, questions were raised whether asking manufacturers of the vaccines to assess potential harms of their own products in which they have huge financial interests can be in the best interest of public health.
It is also questionable, “whether the extreme secrecy, with life-long confidentiality agreements, which the EMA imposed on its working group members and scientific experts, is needed; is legitimate; is in the public interest; and guarantees that the administration enjoys legitimacy.”[4]
Mice for Thought
A study conducted by renown scientists Rotem Inbar, Ronen Weiss, Lucija Tomljenovic, Maria-Teresa Arango, Yael Deri, Christopher A. Shaw, Joab Chapman, Miri Blank and Yehuda Shoenfeld examined the behavioural abnormalities in young female mice following the administration of aluminum adjuvants and the human papillomavirus (HPV) vaccine Gardasil®.
Interestingly, the study concluded that both aluminum and the Gardasil® vaccine brought about behavioural as well as cognitive abnormalities in mice.
Therefore, given the strong effects seen in mice, there is a great need for further examination of the adverse effects of this heavily advertised vaccine. The inconsistency found with regards to HP-vaccine safety as well as the results found in this study should alert public health officials. The scientists involved in the so-called Inbar-study highlight “the necessity of proceeding with caution with respect to further mass-immunization practices with a vaccine of yet unproven long-term clinical benefit, which is capable of inducing immune-mediated cross-reactions with neural antigens of the human host”[5]. Moreover, they recommend new guidelines and the use of appropriate placebos in future vaccine safety trials.
Conspiracy or Cover-up
The paper by Inbar et al. gave unprecedented insights into the possible effects of the Gardasil® vaccine as well as aluminum as an adjuvant. It had been entered in September 2015 for publication in the reputable journal “Vaccine”, specialised in the publication of vaccine related news and studies. The study was first accepted on the 31. December 2015 and then later withdrawn by Editor in Chief Gregory A. Poland himself. The official statement read that the reasons for the withdrawal were “due to serious concerns regarding the scientific soundness of the article.”[6].
However, one could easily be mislead into believing that the reasons were of a less scientific nature when checking Poland’s financial ties with Big Pharma.
From Aug. 2013 to Dec. 2014 the Editor in Chief received $17,351 in direct payments from Novartis Vaccines and Diagnostics Inc. and Sanofi Pasteur Inc., most of which was paid for “promotional speaking”[7]. Furthermore, in 2012 and 2013 the Mayo Clinic and various Mayo Foundations, with Poland as listed practitioner, received some $61,475 in payments by Merck for both consulting and research[8]. The exact amounts paid to Poland himself remain undisclosed. However, his close ties to Gardasil® manufacturer Merck certainly do raise eyebrows. The aforementioned figures are based on the voluntary disclosures of pharmaceutical companies alone and do not include possible undisclosed payments made.
Could this have somehow influenced his decision to withdraw the study that shows a causal relationship between the Gardasil® vaccine and behavioural abnormalities in young female mice?
Money vs Health
When huge financial interests are at stake, critical voices are often “overheard” and discredited, no matter how reputable they may be or have been. Hence, despite its revealing results and undoubtedly great importance, there appears to be “some” reason the public is not to know about the results of the Inbar-Study.
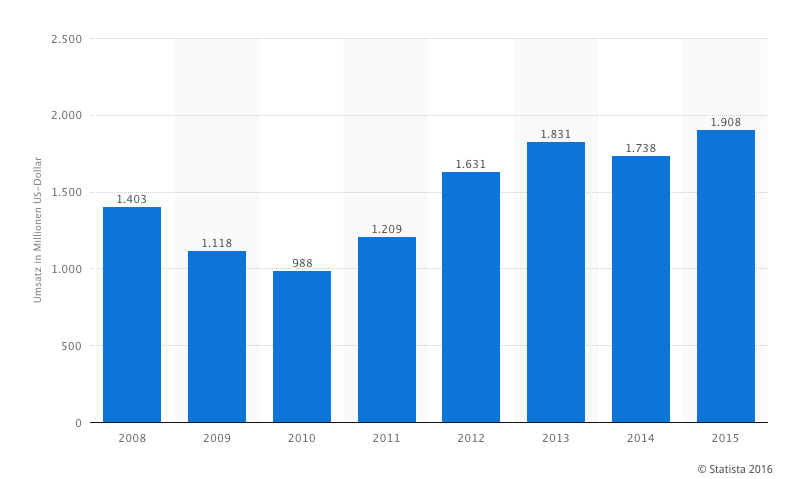
Taking the close financial ties of Poland with Merck and other vaccine makers into account, the intent of the withdrawal is rather blurry. Yet taking a look at Merck’s 1.908 billion dollars revenue in 2015[9] (tendency rising) with Gardasil® alone perhaps provides the missing piece of the puzzle we all deserve to know about.
So can we blindly trust companies that are making a profit from vaccines to be in charge of their safety?
Do not wait for others to take responsibility and report any suspected or proven damages from vaccines to the local authorities in charge!
USA
Vaccine Adverse Event Reporting System (VAERS)
https://vaers.hhs.gov/esub/index
Europe
European database of suspected adverse drug reaction reports
http://www.adrreports.eu/en/index.html
Germany
Paul-Ehrlich-Institut (PEI) und das Bundesinstitut für Arzneimittel und Medizinprodukte (BfArM)
https://verbraucher-uaw.pei.de/fmi/iwp/cgi?-db=Verbraucher-UAW&-loadframes
—
SOURCES
[1] FDA – http://www.fda.gov/BiologicsBloodVaccines/Vaccines/ApprovedProducts/UCM094042
[2] EMA – Europe – http://www.ema.europa.eu/docs/en_GB/document_library/Press_release/2015/07/WC500189481.pdf
[3] Page 2 – Complaint to the European Medicines Agency (EMA) over maladministration at the EMA – Complaint to the European Medicines Agency (EMA) over maladministration at the EMA
http://nordic.cochrane.org/sites/nordic.cochrane.org/files/uploads/ResearchHighlights/Complaint-to-EMA-over-EMA.pdf
[4] Page 1 – Complaint to the European Medicines Agency (EMA) over maladministration at the EMA – Complaint to the European Medicines Agency (EMA) over maladministration at the EMA
http://nordic.cochrane.org/sites/nordic.cochrane.org/files/uploads/ResearchHighlights/Complaint-to-EMA-over-EMA.pdf
[5] Inbar R, et al. Behavioral abnormalities in young female mice following administration of aluminum adjuvants and the human papillomavirus (HPV) vaccine Gardasil. Vaccine (2016), http://dx.doi.org/10.1016/j.vaccine.2015.12.067
[6] Inbar R, et al. Behavioral abnormalities in young female mice following administration of aluminum adjuvants and the human papillomavirus (HPV) vaccine Gardasil. Vaccine (2016), http://www.ncbi.nlm.nih.gov/pubmed/26778424
[7] Pro Publica – https://projects.propublica.org/docdollars/doctors/pid/194859
[8] Pro Publica – https://projects.propublica.org/d4d-archive/search?utf8=✓&term=Gregory+Arthur+Poland&state%5Bid%5D=&services%5B%5D=&period%5B%5D=&showall=true
[9] Statistica – http://de.statista.com/statistik/daten/studie/316936/umfrage/umsatz-von-merck-and-co-mit-dem-arzneimittel-gardasil/
Picture © Mikkolem | Dreamstime.com